Dear malariacontrol.net user,
In our last posting, we described several updates and improvements we`ve made to our model. This round, we`d like to report on a research question we`ve addressed with the model: what is the optimal strategy for distributing a malaria vaccine?
The question is particularly relevant since one promising vaccine—RTS,S—is currently undergoing phase III clinical trials (the last stage of testing before licensing) in infants and children in seven African countries.
As you know, malaria transmission can be reduced by spraying insecticide to reduce the number of mosquitoes, and by reducing the number of bites by sleeping under insecticide-treated bed nets. Treating patients with anti-malarial drugs also reduces transmission because it reduces the chance that a mosquito will become infected with an active malaria parasite when it bites the patient. For the same reason, a malaria vaccine could reduce the overall malaria burden, both by protecting an individual from disease, but also because overall transmission will be reduced: that is, mosquitoes who bite the vacinated individual won`t become infected with the malaria parasite through that bite.
Assuming the RTS,S vaccine fulfills its promise and is licensed, the next question will be how to best distribute the vaccine. One possibility would be to include the malaria vaccine in the suite of immunizations planned through the World Health Organization's Expanded Programme on Immunization (EPI), which aims to provide universal access to immunization against several infectious diseases during the first three months of life. Another possibility would through a mass malaria vaccination campaign, or some combination of both ideas.
The malaria model developed and calibrated though your efforts is the ideal tool to address this problem. Researchers here at the Swiss Tropical and Public Health Institute submitted an ensemble of models of P. falciparum dynamics to assess the likely public health impact of the RTS,S malaria vaccine to malariacontrol.net; your machines did the rest.
These models included different assumptions about the decay of acquired immunity against malaria, different transmission efficiencies, and, critically, patient access to treatment. The passage of thousands of hypothetical individuals through different stages of malaria infection was simulated; movement between stages occurs stochastically (by chance) at a probability based on field data. Each model was used to predict the health benefits during 14 years of RTS,S deployment through four different immunization strategies:
(1) EPI without catch-up vaccination for infants who were not immunized during their first three months of life
(2) EPI with catch-up vaccination for infants who were not immunized during their first three months of life
(3) EPI and supplementary vaccination of school children
(4) Mass vaccination campaigns every five years
The result
The predicted benefits of EPI vaccination programs over the 14-year period were modest and similar over a wide range of settings. However, there were differences depending on the malaria transmission levels, or entomological inoculation rate [EIR].
Strategy number 2 (EPI with an initial catch-up phase) averted the most deaths per vaccine dose at higher EIRs (11 and 20 infectious bites per person per year).
At the lowest EIR (2 infectious bites per year), strategy 4, (mass vaccination) substantially reduced transmission, leading to much greater health improvement per dose than other strategies, even at modest coverage.
You can see this difference in the lower right figure on the graphic below:
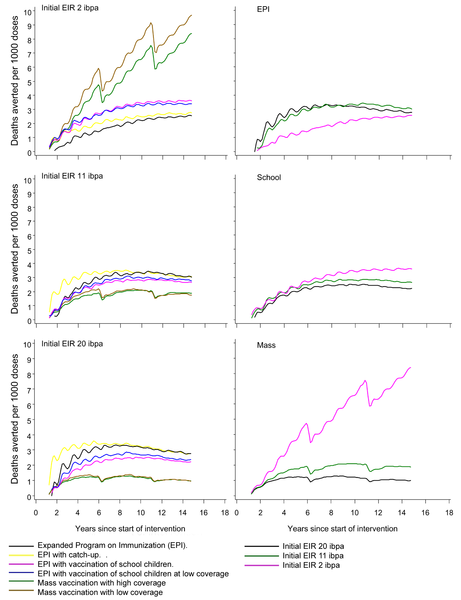
The conclusion: targeted mass vaccination with RTS,S in low transmission settings may have greater health benefits than vaccination through national EPI programs.
Most importantly, the computer-intensive approach provides more secure predictions than can be obtained using any single model. – this computer intensive approach was only possible through our combined efforts. You can read more about the work at
Ensemble Modeling of the Likely Public Health Impact of a Pre-Erythrocytic Malaria Vaccine
As always, thank you for your support and collaboration!
Still to come…
Of course, one question leads to another. The next step will be to combine the outputs of multiple models with economic analyses to provide a rational basis for the design of vaccine-containing malaria control and elimination programs. Stay tuned for more work units!
Mariah
____________
Mariabeth Silkey
Swiss Tropical and Public Health Institute
http://www.swisstph.ch
- Reply
- Quote
Thank you for this update.
It is good to have an insight into what happens to our crunching.
____________
Warped

- Reply
- Quote
Thank you for this update.
It is good to have an insight into what happens to our crunching.
I TOTALLY agree!!! Thanks from me too!!!
____________

- Reply
- Quote
It's a great update !!!
Thank you for that ! I was looking for this kind of message to come back to your project !
- Reply
- Quote
It's a great update !!!
Thank you for that ! I was looking for this kind of message to come back to your project !
I'm agry with my friend Dudumomo!
____________
- Reply
- Quote
Dear Mariah,
Thank you very much for an interesting, informative and understandable report. Very much appreciated.
John.
- Reply
- Quote
Very nice to read news from the research team.
- Reply
- Quote
Many thanks for the encouraging update.
John
____________
- Reply
- Quote
Dear malariacontrol.net user,
Some of you have been having a bit of trouble with some jobs not completing properly. Thanks for letting us know right away! Michael T. has sorted out the lost computing credits, and he's re-setting the initial values for those models and sending them out for another try at fitting.
These things happen on the frontiers of science. If we knew what to expect every time we run a model, we wouldn't need to run the experiment! In this case, it's all due to re-fitting a model parameter for survival, sigma2i. The survival factor grew too fast and sent the density of infection reeling towards infinity. For those of you unlucky enough to download those particular model runs, the models never converged... and, as you know, the work unit never completed.
Why refit the models? We were investigating a wider parameter space for the model fitting than we had earlier. Think of the parameter space as a complicated roller coaster path with two or three valleys. Depending on where you start the search for the minimum value, you'll land in one or another valley. The key is to keep searching across the entire parameter space to make sure you've found the true, overall, minimum. It was in this search, for the global minimum of sigma2i, that our fitting algorithm got stuck.
In our last post, we talked about how your simulations helped us answer a question about the optimal strategy for distributing a malaria vaccine. This time around we'd like to tell you about another problem we looked at with your model runs - an analysis to see which factors were most important to determine the effective lifetime of long-lasting insecticide treated nets.
Long-lasting insecticide treated nets work: we know they reduce malaria transmission by protecting people from infectious bites. But even more important from a malaria control perspective, the nets keep the mosquitos from getting malaria from infected humans. In short, how long can we expect the epidemiological effect of these long-lasting nets to last?
We ran simulation models to explore the influence of pre-intervention transmission level, initial coverage, net attrition, and both physical and chemical net decay. The effective lifetime was most sensitive to the pre-intervention transmission level, with a lifetime of almost 10 years at an entomological inoculation rate of two infectious bites per adult per year, but of little more than 2 years at 256 two infectious bites per adult per year. The LLIN attrition rate and the insecticide decay rate were the next most important parameters. The lifetime was surprisingly insensitive to physical decay parameters, but this could change as physical integrity gains importance with the emergence and spread of insecticide resistance.
The strong dependency of the effective lifetime on the pre-intervention transmission level indicated that the required distribution frequency may vary more with the local entomological situation than with quality of the net or the characteristics of the distribution system. This highlights the need for malaria monitoring both before and during intervention programmes, particularly since there are likely to be strong variations between years and over short distances.
For more information on this result, including some pretty pictures, see http://www.malariajournal.com/content/11/1/20/abstract
So, well done you, malaria control net contributor!
As always, thank you for your support and collaboration.
Mariah Silkey
____________
Mariabeth Silkey
Swiss Tropical and Public Health Institute
http://www.swisstph.ch
- Reply
- Quote
and, as you know, the work unit never completed.
Never? I was leaving one running in the hope that late results would be better than none at all. Have we any way of telling if a WU will eventually reach an endpoint?
- Reply
- Quote
